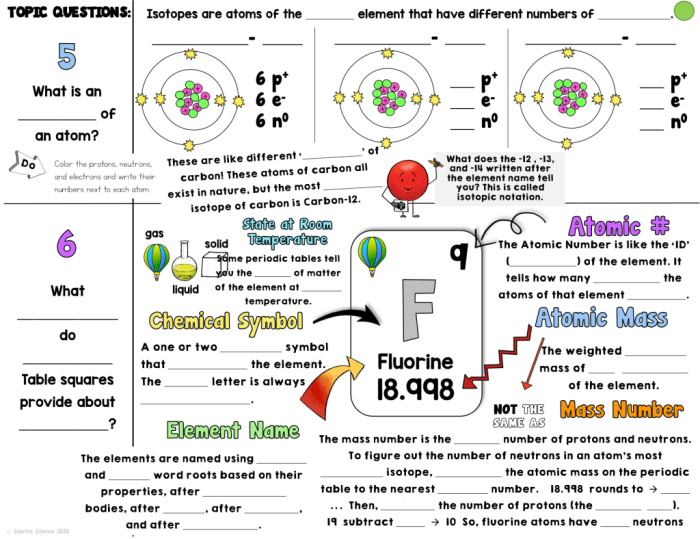
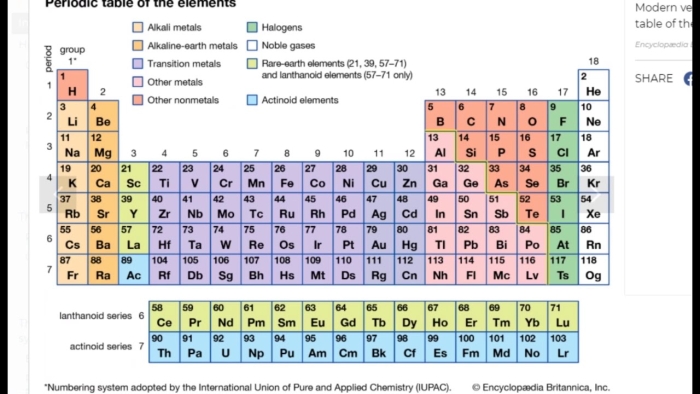
The periodic table sits in nearly every chemistry classroom, yet most fifth grade students see it as an intimidating grid of letters and numbers. Once you understand how to read it, though, the periodic table becomes one of the most useful tools in science. It organizes all known elements in a way that reveals patterns about how they behave and interact with each other.
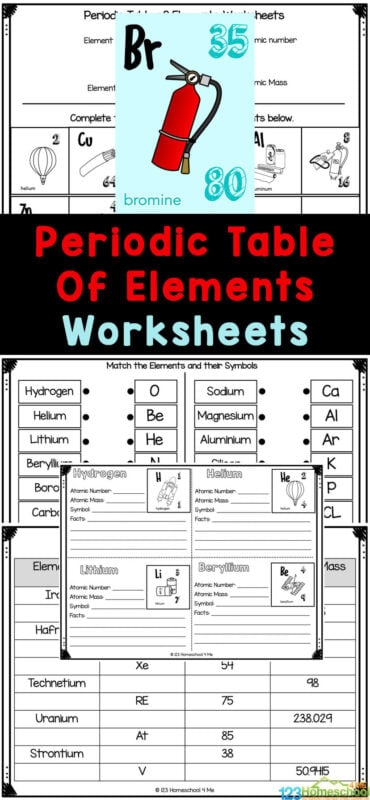
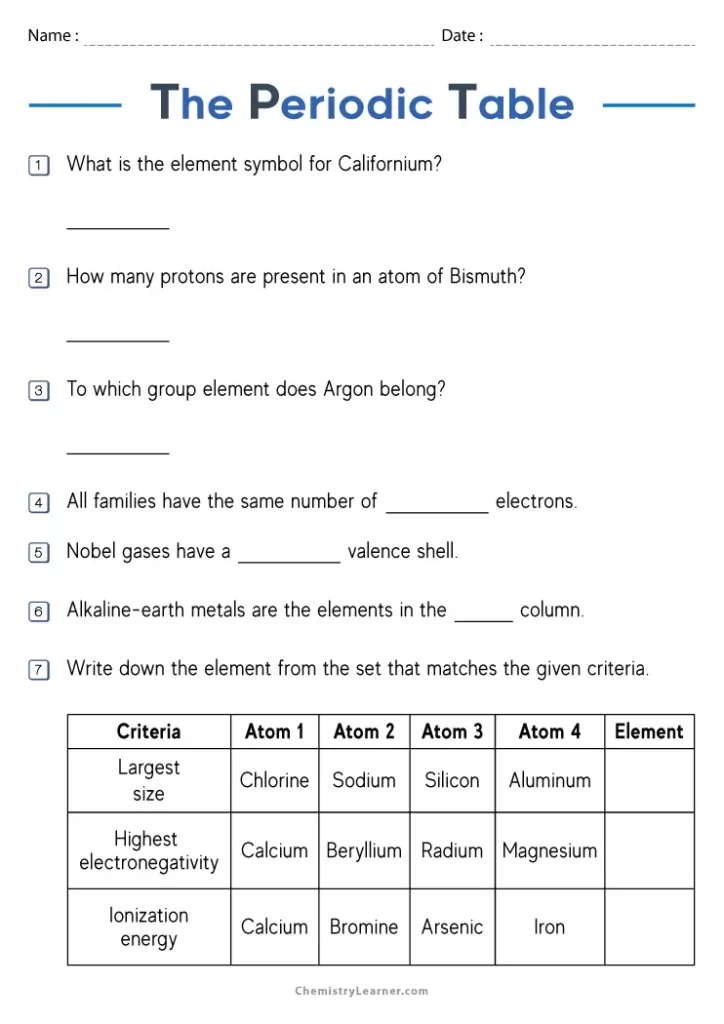
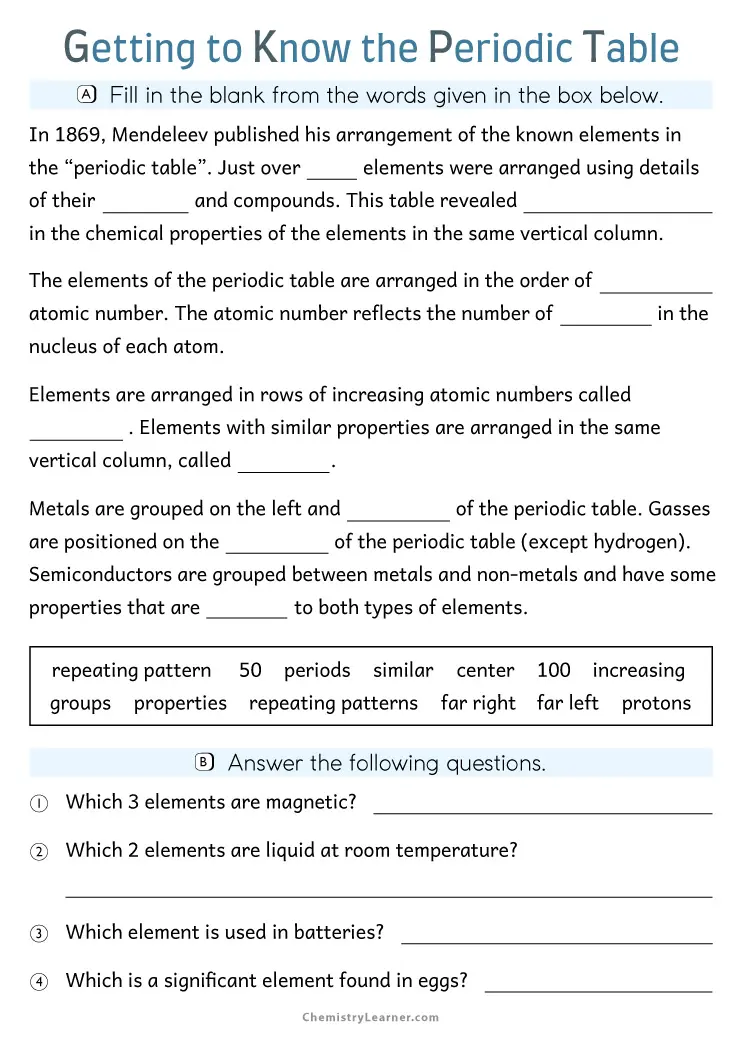
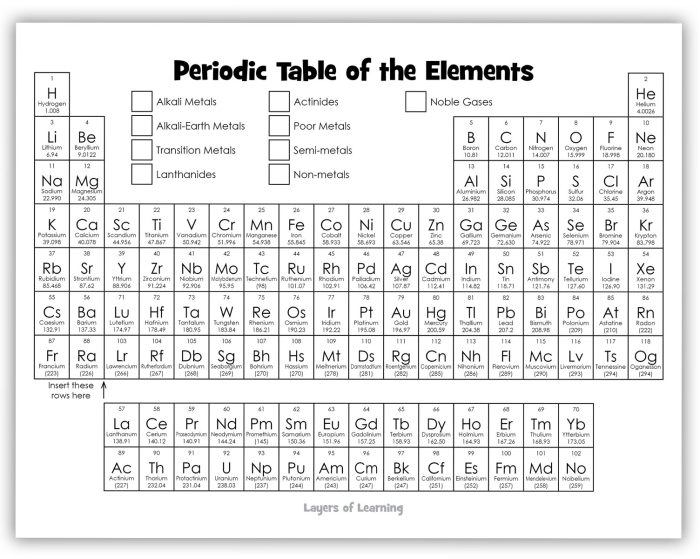
Each box on the periodic table contains several pieces of information about an element. The symbol, typically one or two letters, appears in the center of the box. Above it sits the atomic number, which tells you how many protons are in an atom of that element. Below the symbol, you’ll find the atomic mass, a number that represents the average weight of the element’s atoms. These numbers and counting systems form the foundation for understanding chemistry at any level.
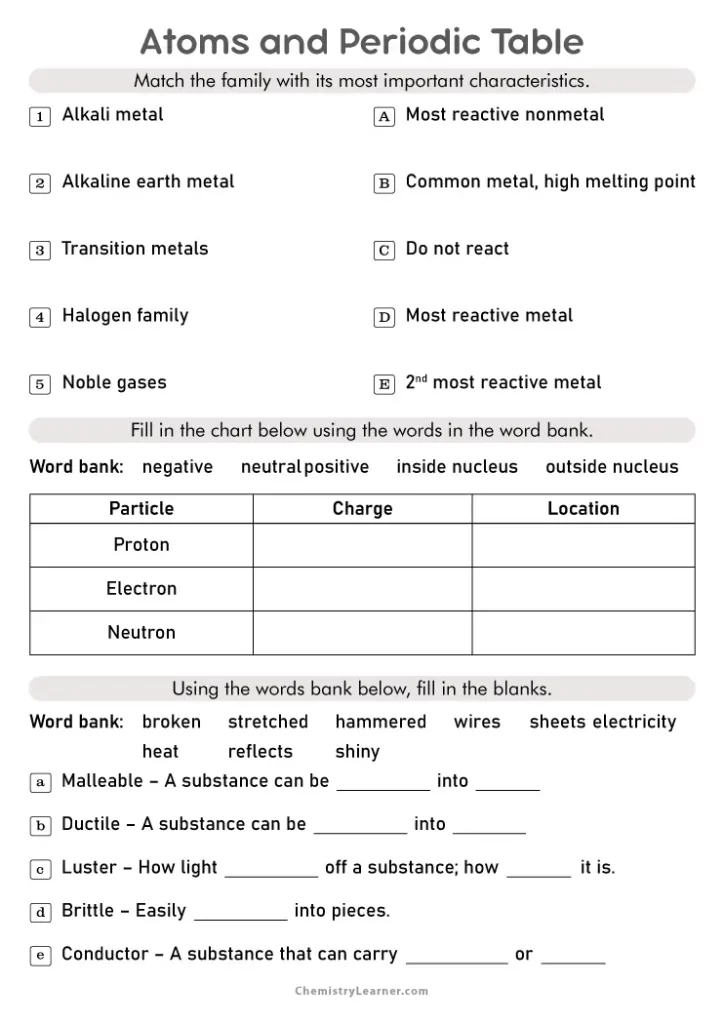
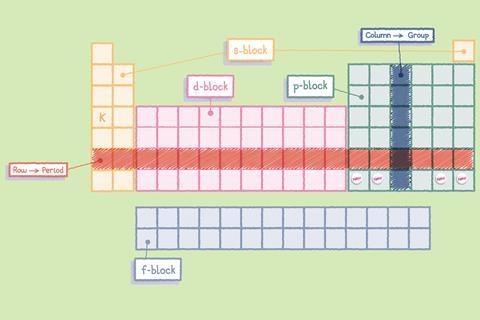
The arrangement of elements matters just as much as the information in each box. Elements are organized by rows, called periods, and columns, called groups. Elements in the same group share similar chemical properties because they have the same number of electrons in their outer shell. Hydrogen and helium occupy the first period, while heavier elements like gold and uranium sit further down the table.
Learning to navigate the periodic table opens doors to understanding how materials work. When you recognize patterns in how elements are positioned, you can predict how they might react with other elements. This skill connects directly to practical applications like calculating volume of chemical compounds or solving problems involving atomic structure.
For fifth grade students beginning their chemistry journey, starting with the most common elements like carbon, oxygen, and nitrogen makes sense. These elements appear frequently in nature and in your own body, making them relatable and easier to remember. With practice using reference materials and worksheets, reading the periodic table becomes second nature.
Worksheet Practice Section